After John Oliver’s Last Week Tonight episode aired on 3rd of July, people started questioning the authority, not just of their healthcare workers, but laws regarding healthcare as well.
It is good to question things and not buy into concepts or follow other’s opinions blindly, but there are times when you need to be certain and know that you can place your trust in the hands of the authority. Healthcare and law are certainly one of these cases.
Unfortunately, we’ll need to second-guess these authorities in order to change the system into the one we can trust again.
The Medical Device Industry

By the analysis conducted in 2017, the United States represents 40 percent of the global medical device market. This means that it remains the world’s largest market, with the medical device industry worthy around $156 billion. By 2024, the market is expected to grow to $208 billion.
The obvious problem arises concerning quality control. Many of these devices are not being adequately tested, which is to be expected since there are more than 6,500 medical device companies in the United States.
There is a hyperproduction in this field that needs to sell their products, and, as John Oliver pointed out, we are therein being treated as guinea pigs. Devices are proved to work only through time, but by that time, thousands of these devices are being distributed and implanted.
Notorious Case
This is the case with the notorious IVC filters used to prevent deep vein thrombosis from interfering with lungs. In 2010, the FDA issued a safety warning regarding the adverse issues that have been reported over the five-year period. These included device migration, fracture and further thrombosis.
Even though the overuse in IVC filters declined since 2010, the retrieval of these harmful devices remains low, which is the main cause of their malfunction. The rate of implantations is still 25 times higher than in Europe, with 250,000 IVC filters surgically implanted each year in the U.S.
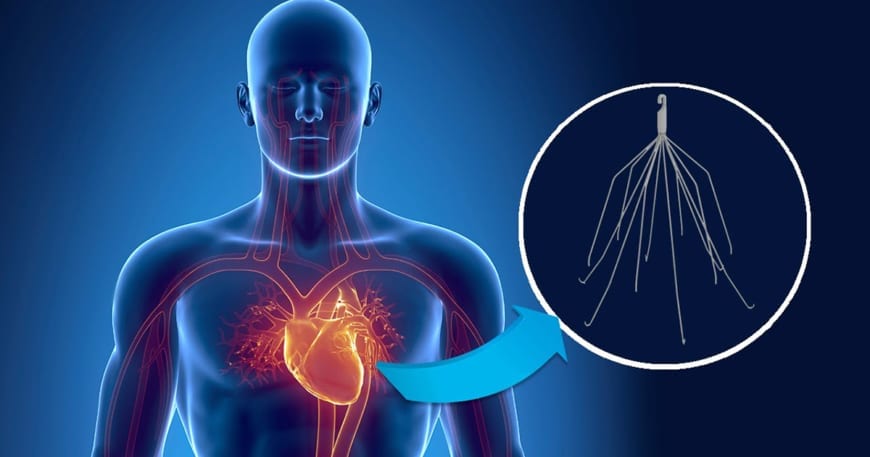
There are more than 10,000 lawsuits filed across the country, mostly against two big manufacturers, Bard Medical and Cook Medical. Although they were hit with verdicts, both companies won the defense verdicts in other cases, so additional trials were scheduled for this year, hopefully leading to settlement and resolution. For more information and a free consultation on this topic, click here.
Better Safe Than Sorry
We never cared to investigate the meaning of the labels ‘FDA approved’ and ‘FDA cleared’. Those that watched Last Week Tonight’s episode know that there is a huge difference between the two. And in both cases, the certificate does not ensure safety.
So what’s the difference?
Products that can cause serious injury or illness need to undergo FDA approval procedure, however, this doesn’t mean that the inquiry and testing are conducted by the FDA itself. The FDA only checks the results, that are usually not the products of rigorous testing as you’d expect.
A product is FDA cleared if it proves to be substantially equivalent to a predicate, predicate being another similar device that has already been cleared. However, some of these predicates are in time proved to be defective, but devices leaning on their credence are still considered cleared. This kind of unreliable testing of medical devices resulted in more than 80,000 deaths since 2008.
We should start wondering how it got to so many unchecked devices being out in the market, without even the doctors and surgeons knowing they cause serious damage. We can’t change the system overnight, but we can start by being more cautious and thinking twice before deciding whether our trust is in the right hands.
Unfortunately, healthcare is not provided, it is sold. This requires us to be more aware of the market we buy into and consider its interplay with other, seemingly unrelated structures such as healthcare and law.